News Center
Popular products
Sales Department Tel:
0512-52836238
0512-52836128
0512-52358728
Sales Department Fax
0512-52836278
Sales Department Email
xu@worldbrom.com
Purchasing Department Tel/ Fax
0512-52836228
Headquarters Address US Office
, No. 18 Haitian Road, Advanced Material Industrial Park, Changshu City, Jiangsu
Tel
+1 832-857-1028
U.S. office mailbox
Lauren@worldbrom.com
shelia@worldbrom.com
U.S. office address
Creekside Park, The Woodlands, Texas 77375, USA
corporate website
www.worldbrom.com
0512-52836238
0512-52836128
0512-52358728
Sales Department Fax
0512-52836278
Sales Department Email
xu@worldbrom.com
Purchasing Department Tel/ Fax
0512-52836228
Headquarters Address US Office
, No. 18 Haitian Road, Advanced Material Industrial Park, Changshu City, Jiangsu
Tel
+1 832-857-1028
U.S. office mailbox
Lauren@worldbrom.com
shelia@worldbrom.com
U.S. office address
Creekside Park, The Woodlands, Texas 77375, USA
corporate website
www.worldbrom.com
Procurement Hotline
0512-52836128
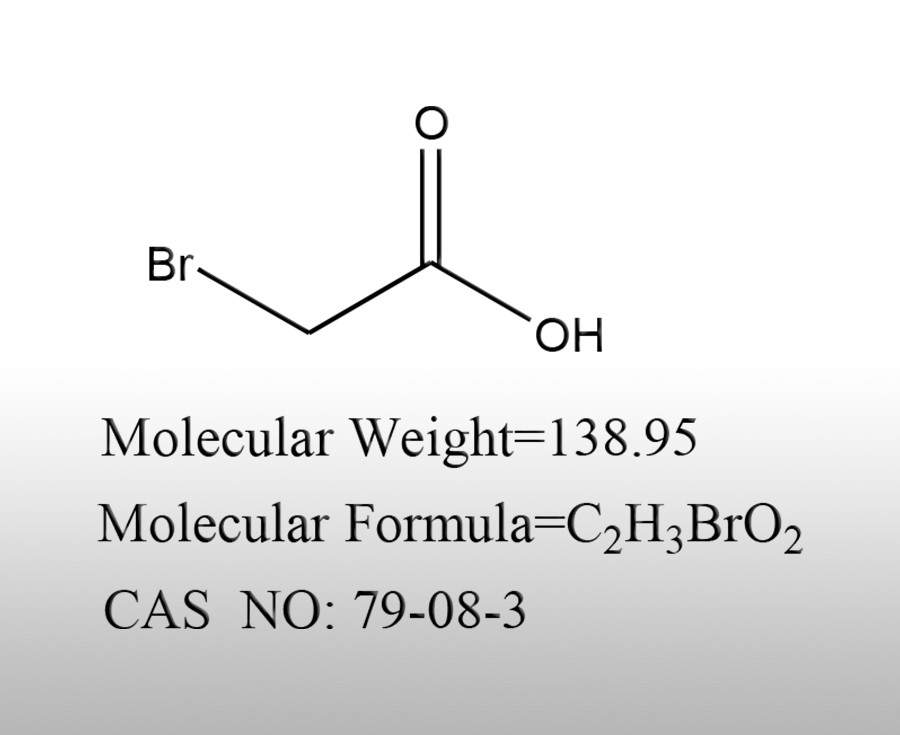
What are the applications of bromoacetic acid in the materials industry
Bromoacetic acid (CAS: 79-08-0) is a highly active halogenated carboxylic acid, which contains both carboxyl (- COOH) and bromine (- Br) reactive sites in its molecular structure. It can undergo nucleophilic substitution reactions and participate in various chemical reactions such as esterification and carboxymethylation. It is a key chemical intermediate for material synthesis and modification. It is not directly used as a finished product in the materials industry, but as a monomer, modifier, functionalizing reagent, crosslinking agent, etc., participating in the preparation of various functional materials, widely covering polymer materials, water treatment materials, new energy materials, coating adhesives, functional dye assistants and other fields. The following is a complete analysis of the segmented application scenarios.Sources:www.worldbrom.com | PublishDate:2026.03.10
1、 Synthesis and Modification of Functional Polymer Materials
Bromoacetic acid is a core reagent for the functionalization modification of polymer materials. It can introduce carboxyl and bromine groups into ordinary polymer chains through grafting, copolymerization, cross-linking, and other methods, endowing the material with special properties such as water solubility, reactivity, and pH sensitivity, and expanding the application boundaries of polymer materials.
It can be copolymerized with vinyl monomers, diene monomers, etc. to prepare functional copolymers containing carboxyl and bromine groups. These copolymers have adjustable polarity and excellent solubility, and can be further processed as polymer substrates. Meanwhile, bromoacetic acid is often used as a carboxymethylation reagent to graft modify natural or synthetic polymers such as cellulose, chitosan, polyvinyl alcohol, etc., improving the hydrophilicity, adhesion, and mechanical strength of the materials, and adapting them to fields such as biomedical materials and intelligent response materials. Among them, pH sensitive polymer materials synthesized with the participation of bromoacetic acid can undergo swelling or shrinkage response according to the external acid-base environment, and can be used as drug controlled release carriers, intelligent sensing materials, etc., with outstanding potential applications in the fields of biomaterials and medical consumables.
2、 Ion exchange resin and water treatment functional materials
Ion exchange resin is a core material for water treatment and industrial separation and purification. Bromoacetic acid plays a key modifying role in its synthesis process and is an important additive for improving the ion exchange capacity and adsorption performance of resins. It is mainly used in the preparation of anion exchange resins and chelating resins.
Through nucleophilic substitution reaction, bromoacetic acid can correctly introduce functional groups such as carboxyl and quaternary ammonium salts into the resin skeleton, greatly improving the resin's adsorption and exchange capacity for metal ions and anions and cations, optimizing the resin's exchange rate and regeneration performance. The ion exchange resin modified with bromoacetic acid can efficiently remove heavy metal ions, calcium and magnesium ions, and organic impurities from water. It is widely used in industrial pure water preparation, sewage treatment, heavy metal recovery, food and beverage water purification, and other scenarios. It is also suitable for material separation and purification in the chemical and pharmaceutical industries, and is an indispensable synthetic intermediate for high-performance water treatment materials.
3、 Functional coatings, adhesives, and sealing materials
In the field of coatings and adhesives, bromoacetic acid is mainly used as a reactive modifier and crosslinking agent to optimize the adhesion, corrosion resistance, weather resistance of coatings, as well as the bonding strength and stability of adhesives. It is suitable for sub sectors such as industrial protection, water-based coatings, and special adhesives.
Introducing bromoacetic acid into the water-based coating system can improve the water solubility and film-forming property of the coating, enhance the adhesion between the coating and metal, glass, wood and other substrates, and at the same time give the coating a certain acid and alkali resistance and anti-aging performance, which is suitable for water-based industrial coatings, anti-corrosion coatings, paper surface treatment coatings, etc. In the preparation of adhesives, bromoacetic acid participates in the cross-linking reaction of adhesive molecular chains, enhancing the cohesive strength and water resistance of the adhesive layer. It is particularly suitable for water-based adhesives, special composite adhesives, and can be used in wood processing, packaging materials, fabric composites, and other scenarios to improve the pain points of traditional adhesives such as easy delamination and poor weather resistance.
4、 New Energy Storage Battery Materials (Electrochemical Materials)
As a new type of electrochemical additive, bromoacetic acid has achieved breakthrough applications in the field of new energy materials such as zinc iodine batteries and aqueous energy storage batteries. It is a key functional additive for improving battery reversibility and cycle life, and belongs to the forefront application direction of energy storage materials.
In the zinc iodine battery system, the bromoacetate ions generated by the dissociation of bromoacetic acid can bind with iodine ions through strong electrostatic interactions, suppress the hydrolysis side reaction of iodine ions, stabilize the redox pairs of the battery, and regulate the deposition behavior of the zinc negative electrode, alleviating the problems of zinc dendrite growth and negative electrode corrosion. The battery system modified with bromoacetic acid has significantly improved reversibility, and can maintain high capacity retention even after thousands of cycles at high current density, effectively improving the cycle life and energy density of water-based energy storage batteries. It helps in the research and industrialization of low-cost and high safety energy storage materials, and is suitable for new energy application scenarios such as energy storage power stations and portable electronic devices.
5、 Functional dyes, pigments, and textile material additives
Bromoacetic acid is used in the field of dyes and textile materials as an intermediate for the modification of dye molecules and the synthesis of functional additives. It is used to prepare high-performance reactive dyes and fabric functional finishing agents, improving the dye's coloring rate, fixation, and textile functionality.
It can react with aromatic amines and heterocyclic compounds, introduce carboxyl and bromine substituents into dye molecules, improve the hydrophilicity and reactivity of dye molecules, and prepare high fixation rate and high color fastness reactive dyes, especially suitable for the printing and dyeing of natural fabrics such as cotton and linen, solving the problems of uneven coloring and easy fading of traditional dyes. Meanwhile, bromoacetic acid can be used to synthesize textile additives such as anti-static agents, softeners, and waterproofing agents. By functionalizing the fiber surface, it endows textiles with additional properties such as anti-static, hydrophilic, and antibacterial, enhancing the added value and market competitiveness of textile materials.