News Center
Popular products
Sales Department Tel:
0512-52836238
0512-52836128
0512-52358728
Sales Department Fax
0512-52836278
Sales Department Email
xu@worldbrom.com
Purchasing Department Tel/ Fax
0512-52836228
Headquarters Address US Office
, No. 18 Haitian Road, Advanced Material Industrial Park, Changshu City, Jiangsu
Tel
+1 832-857-1028
U.S. office mailbox
Lauren@worldbrom.com
shelia@worldbrom.com
U.S. office address
Creekside Park, The Woodlands, Texas 77375, USA
corporate website
www.worldbrom.com
0512-52836238
0512-52836128
0512-52358728
Sales Department Fax
0512-52836278
Sales Department Email
xu@worldbrom.com
Purchasing Department Tel/ Fax
0512-52836228
Headquarters Address US Office
, No. 18 Haitian Road, Advanced Material Industrial Park, Changshu City, Jiangsu
Tel
+1 832-857-1028
U.S. office mailbox
Lauren@worldbrom.com
shelia@worldbrom.com
U.S. office address
Creekside Park, The Woodlands, Texas 77375, USA
corporate website
www.worldbrom.com
Procurement Hotline
0512-52836128
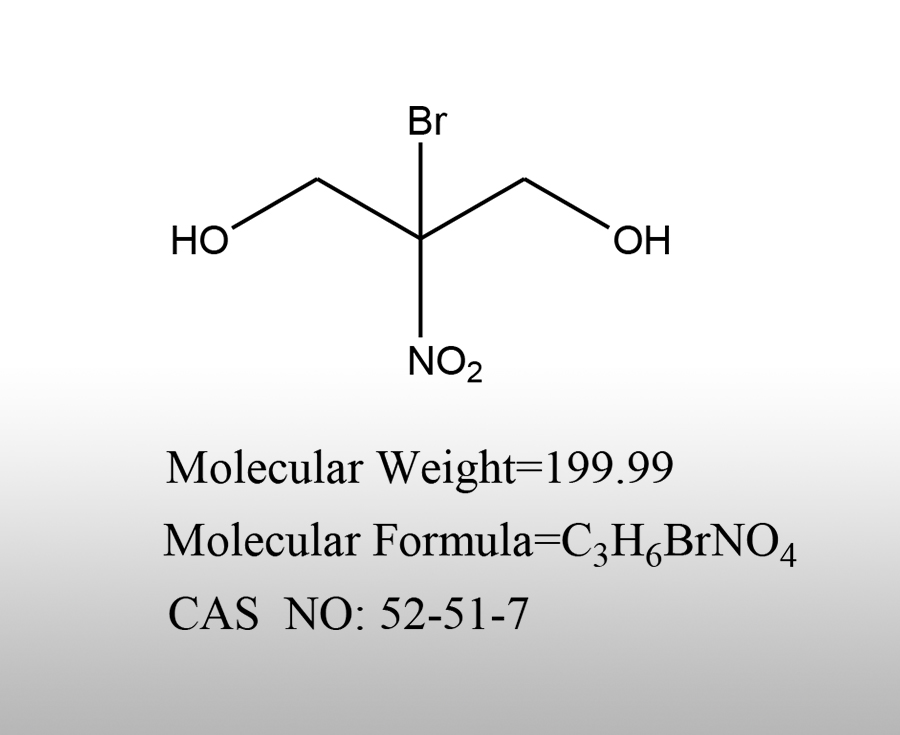
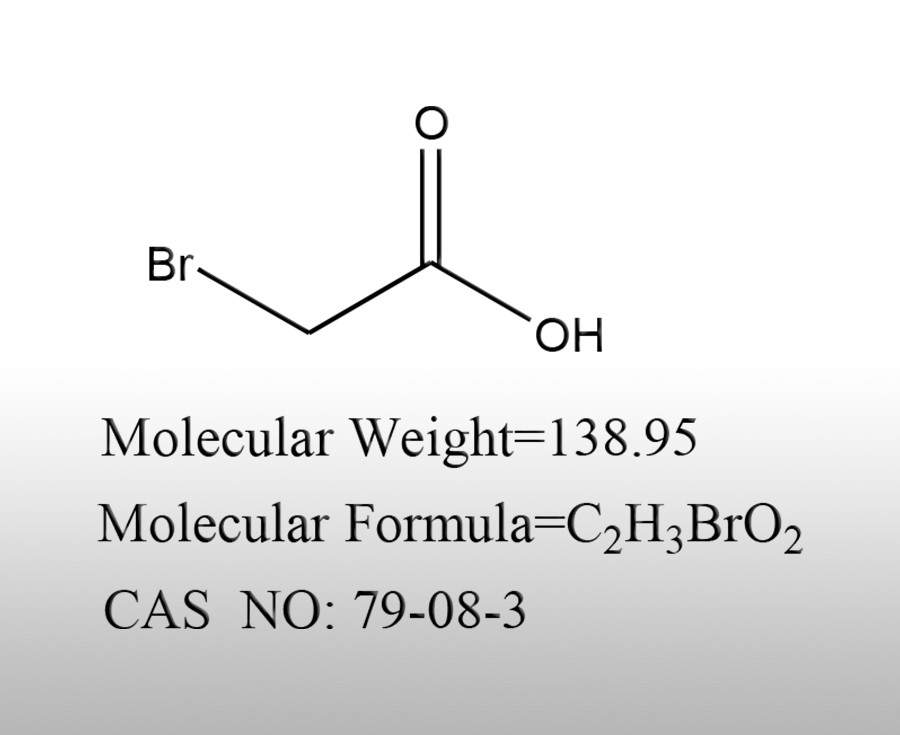
What are the characteristics of bromoacetic acid
Bromoacetic acid (BrCH ₂ COOH, CAS 79-08-3) is a highly active, corrosive, and toxic halogenated acetic acid intermediate. Its core characteristics can be summarized as strong acidity, high reactivity, strong hygroscopicity, strong corrosiveness, high toxicity, and multi-purpose intermediate. The official characteristics are systematically explained from four dimensions: physical and chemical, chemical, safety, and applicationSources:www.worldbrom.com | PublishDate:2026.04.14
1、 Physical and chemical properties
Appearance and condition: It is a colorless to slightly yellow hexagonal crystal solid at room temperature, with a strong pungent odor. The pure product is close to colorless, and the industrial product has a slight light yellow color.
Melting point: 49-51 ℃ (normal pressure), easily semi melted due to moisture absorption at room temperature.
Boiling point: 206-208 ℃ (atmospheric pressure), high temperature easily decomposes and releases toxic fumes such as hydrogen bromide (HBr).
Density: 1.934 g/cm ³ (50 ℃), heavier than water.
Solubility: Highly soluble in water, ethanol, ether, soluble in most polar/weakly polar solvents such as acetone, benzene, tetrahydrofuran, etc. The aqueous solution is strongly acidic.
Moisture absorption: extremely strong, rapidly deliquescent, hygroscopic clumping, and even liquefaction when exposed to air, must be strictly sealed to prevent moisture.
Stability: Stable at room temperature, but prone to degradation when exposed to light, heat, and moisture; Cannot coexist with strong oxidants or strong bases; Flammable, can ignite when exposed to open flames or high heat, and decompose to produce toxic gases such as CO, CO ₂, HBr, etc.
Acidity: pKa ≈ 2.86 (25 ℃), much stronger than acetic acid (pKa ≈ 4.76), belonging to medium strong acid, significantly more corrosive than ordinary carboxylic acids.
2、 Chemical properties (highly active bifunctional core)
The molecule contains both carboxyl groups (- COOH) and alpha bromine atoms (- Br), and the dual active sites endow it with multiple reactivity:
Strong acidity reacts with carboxyl groups
Typical carboxylic acid reactions can occur, including salt formation, esterification, acylation, amidation, decarboxylation, etc., to generate derivatives such as bromoacetate, amide, and acyl halide, which are important carboxymethyl building blocks.
High activity of α - bromine atom (strong alkylation)
Bromine is a good leaving group and easily undergoes nucleophilic substitution (SN2). It is a typical strong alkylating reagent that can react rapidly with amines, alcohols, thiols, phenols, enolates, etc. to achieve C -, N -, O -, S-carboxymethylation. It is widely used for carbon chain elongation and heteroatom functionalization.
Hydrolysis and elimination
Under humid or alkaline conditions, it is easily hydrolyzed into glycolic acid (hydroxyacetic acid); Under high temperature/strong alkaline conditions, hydrogen bromide can be removed to generate unsaturated carboxylic acids, with mild reaction conditions and high conversion rates.
Metal corrosiveness
Water solution has strong corrosion on most metals such as iron, copper, aluminum, zinc, etc., generating bromide salts. Storage and transportation require materials such as glass, ceramics, polytetrafluoroethylene, and stainless steel.